Moderna’s next generation COVID-19 vaccine candidate tested in clinical trial participants
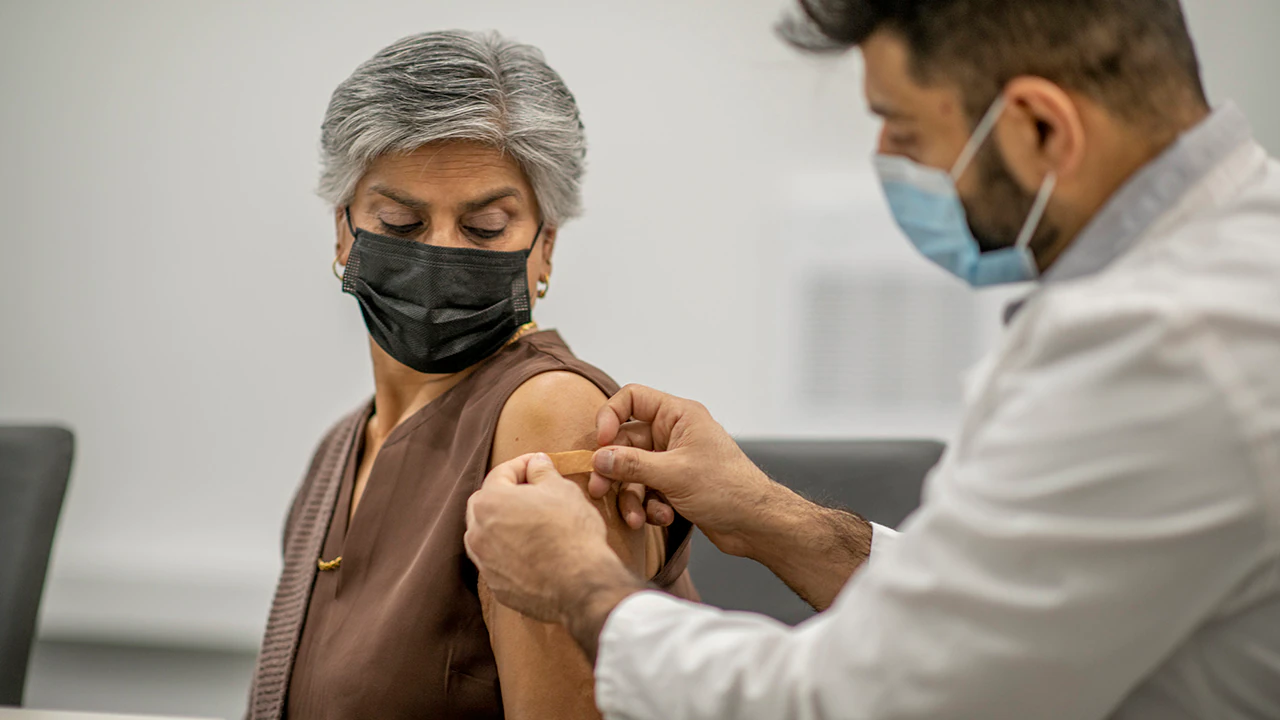
Moderna on Monday announced that it began administering its next generation COVID-19 vaccine candidate in a Phase 1 study. The new candidate, mRNA-1283, can be stored in a refrigerator which could potentially ease storage and shipping hurdles.
“We are pleased to begin this Phase 1 study of our next generation COVID-19 vaccine candidate, mRNA-1283,” Stephane Bancel, Moderna CEO, said in a press release Monday. “Our investments in our mRNA platform have enabled us to develop this next generation vaccine candidate which is a potential refrigerator-stable vaccine that could facilitate easier distribution and administration in a wider range of settings, including potentially for developing countries. We remain committed to helping address this ongoing public health emergency.”
The new clinical trial is evaluating three dose levels of the mRNA-1283, which targets the SARS-CoV-2 spike protein, given to healthy adults in a two-dose series spaced 28 days apart and at one dose level in a single shot.
(iStock)
The currently approved Moderna two-dose vaccine can be stored refrigerated between 2 degrees Celsius to 8 degrees Celsius for up to 30 days prior to use, but cannot be refrozen once thawed. Unpunctured vials may be stored between 8 degrees Celsius and 25 degrees Celsius for up to 12 hours but also cannot be refrozen.
‘PARTIAL’ PFIZER COVID-19 VACCINATION CUTS INFECTION RISK BY 63% AMONG NURSING HOME RESIDENTS: CDC
The new clinical trial is evaluating three dose levels of the mRNA-1283, which targets the SARS-CoV-2 spike protein, given to healthy adults in a two-dose series spaced 28 days apart and at one dose level in a single shot. The results will be compared with the currently authorized dose level in the two-dose series.
CLICK HERE FOR COMPLETE CORONAVIRUS COVERAGE
“mRNA-1283 is intended to be evaluated in futures studies for use as a booster dose for previously vaccinated or seropositive as well as in a primary series for seronegative individuals,” the press release stated.
Source link




